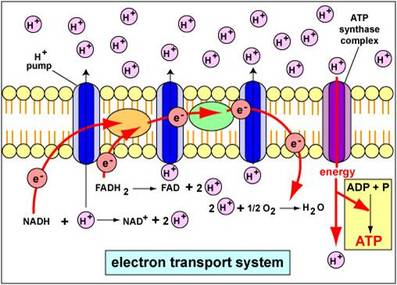
Oxidative phosphorylation is a process by which most ATPs are produced in cellular respiration. Within the inner mitochondiral membrane, there is what is called and Electron Transport Chain/System (ETC/ETS). The ETC involves various enzymes, "complexes", and cytochromes, which act as electron acceptors. Electrons are carried by two main molecules (NADH and FADH2) to the ETC from glycolysis and the Krebs Cycle. Once at the ETC, they give up their electrons as well as hydrogen ions (H+ or protons). The electrons then pass through each enzyme in redox reactions: first becoming reduced, then oxidized. The electrons give off a small amount of energy each time they are transferred as free energy drops. This energy is taken by the proteins in the ETC to pump the protons into the intermembrane space. Eventually the electrons make their way to the bottom of the chain where they meet oxygen (usually).
The proton gradient caused by the high concentration of protons in the intermembrane space begins another process called chemiosmosis. The protons "want" to diffuse back into the mitochondrial matrix and to a lower concentration. A very special enzyme in the membrane called ATP synthase will take protons and transport them into the matrix. In doing so, energy is released and used to phosphorylate ADP into ATP.
The proton gradient caused by the high concentration of protons in the intermembrane space begins another process called chemiosmosis. The protons "want" to diffuse back into the mitochondrial matrix and to a lower concentration. A very special enzyme in the membrane called ATP synthase will take protons and transport them into the matrix. In doing so, energy is released and used to phosphorylate ADP into ATP.
